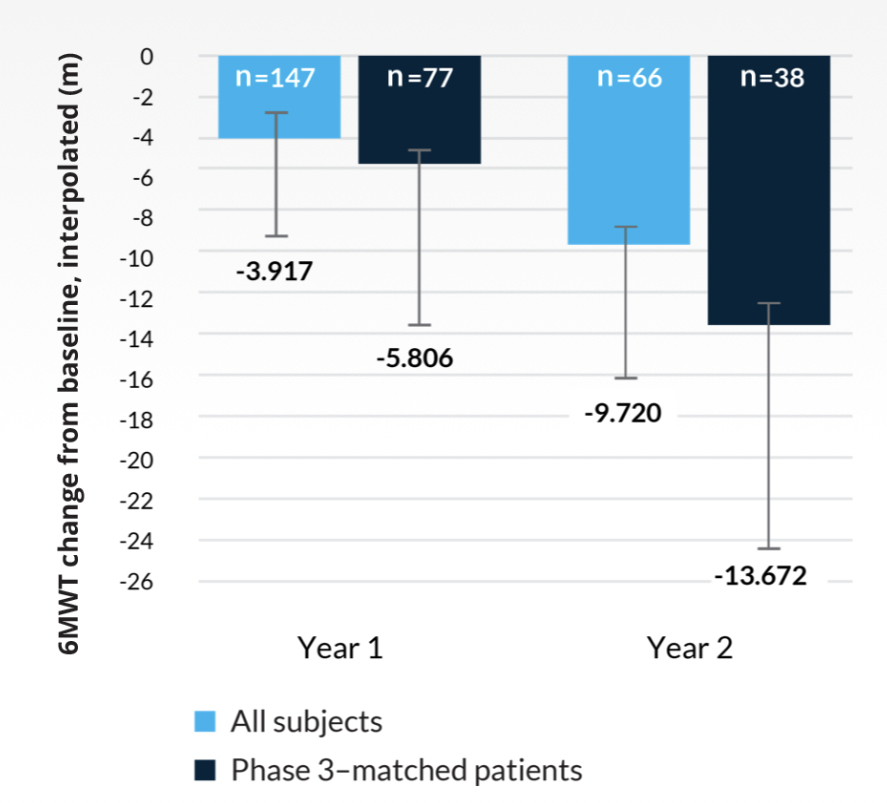
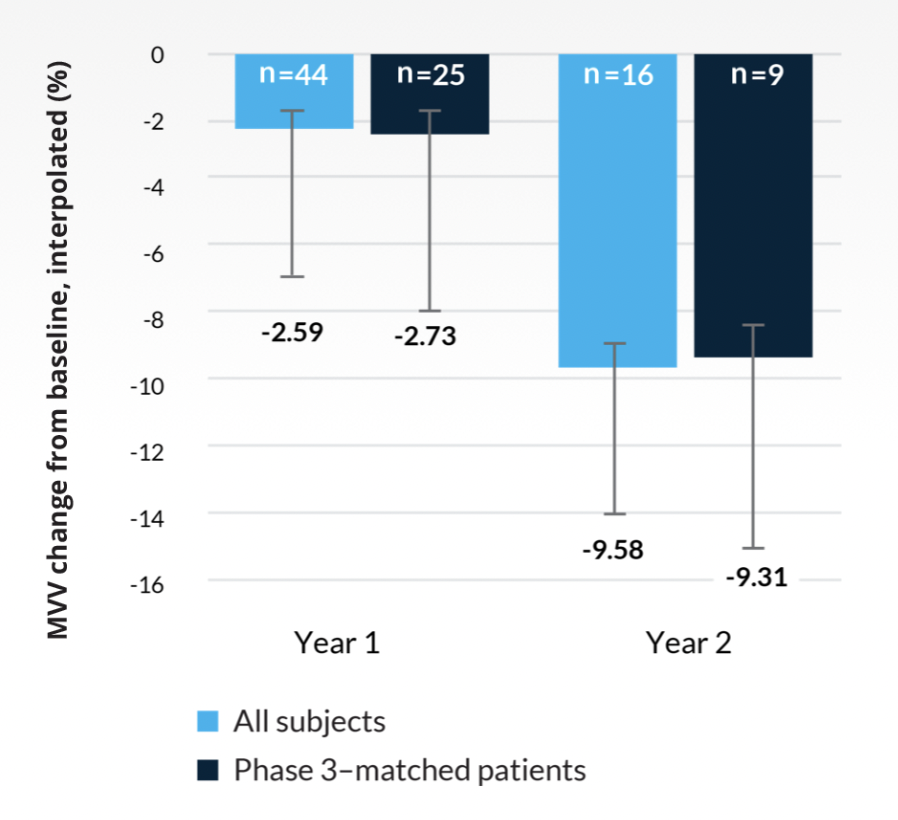
A natural history study of Morquio A demonstrated continued decline in endurance and pulmonary function.2 A subanalysis of the natural history study included patients who met the VIMIZIM® (elosulfase alfa) phase 3 trial inclusion criteria (≥5 years of age with a baseline 6-minute walk test [6MWT] of ≥30 m and ≤325 m).2

Least squares mean 6MWT changes from baseline for Morquio A Clinical Assessment Program (MorCAP) subjects2
Least squares mean maximum voluntary ventilation (MVV) from baseline in patients >14 years2
Life-threatening anaphylactic reactions have occurred in some patients during VIMIZIM infusions. Anaphylaxis, presenting as cough, erythema, throat tightness, urticaria, flushing, cyanosis, hypotension, rash, dyspnea, chest discomfort, and gastrointestinal symptoms in conjunction with urticaria, have been reported to occur during VIMIZIM infusions, regardless of duration of the course of treatment. Closely observe patients during and after VIMIZIM administration and be prepared to manage anaphylaxis. Inform patients of the signs and symptoms of anaphylaxis and have them seek immediate medical care should symptoms occur. Patients with acute respiratory illness may be at risk of serious acute exacerbation of their respiratory compromise due to hypersensitivity reactions and require additional monitoring.
Due to the potential for anaphylaxis, appropriate medical support should be readily available when VIMIZIM is administered and for an appropriate period of time following administration. In clinical trials, cases of anaphylaxis occurred as early as 30 minutes from the start of infusion and up to 3 hours after infusion, and as late into treatment as the 47th infusion.
In clinical trials, hypersensitivity reactions have been observed as early as 30 minutes from the start of infusion but as late as 6 days after infusion. Frequent symptoms of hypersensitivity reactions (occurring in more than 2 patients) included anaphylactic reactions, urticaria, peripheral edema, cough, dyspnea, and flushing.
Because of the potential for hypersensitivity reactions, administer antihistamines with or without antipyretics prior to infusion. Management of hypersensitivity reactions should be based on the severity of the reaction and includes slowing or temporary interruption of the infusion and/or administration of additional antihistamines, antipyretics, and/or corticosteroids for mild reactions. However, if severe hypersensitivity reactions occur, immediately stop the infusion of VIMIZIM and initiate appropriate treatment.
Consider the risks and benefits of re-administering VIMIZIM following a severe reaction.
Patients with acute febrile or respiratory illness at the time of VIMIZIM infusion may be at higher risk of life-threatening complications from hypersensitivity reactions. Careful consideration should be given to the patient’s clinical status prior to administration of VIMIZIM; consider delaying the VIMIZIM infusion.
Sleep apnea is common in MPS IVA patients. Evaluation of airway patency should be considered prior to initiation of treatment with VIMIZIM. Patients using supplemental oxygen or continuous positive airway pressure (CPAP) during sleep should have these treatments readily available during infusion in the event of an acute reaction, or extreme drowsiness/sleep induced by antihistamine use.
Spinal or cervical cord compression (SCC) is a known and serious complication of MPS IVA and may occur as part of the natural history of the disease. In clinical trials, SCC was observed both in patients receiving VIMIZIM and patients receiving placebo. Patients with MPS IVA should be monitored for signs and symptoms of SCC (including back pain, paralysis of limbs below the level of compression, and urinary and fecal incontinence) and given appropriate clinical care.
All patients treated with VIMIZIM 2 mg/kg once per week in the placebo-controlled trial developed anti-drug antibodies. The relationship between the presence of neutralizing antibodies and long-term therapeutic response or occurrence of anaphylaxis or other hypersensitivity reactions could not be determined.
VIMIZIM should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. It is not known if VIMIZIM is present in human milk. Exercise caution when administering VIMIZIM to a nursing mother. There is a Morquio A Registry that collects data on pregnant women and nursing mothers with MPS IVA who are treated with VIMIZIM. Contact MARS@BMRN.com for information and enrollment.
Safety and effectiveness in pediatric patients below 5 years of age have not been established and are currently being evaluated.
In clinical trials, the most common adverse reactions (≥10%) occurring during infusion included pyrexia, vomiting, headache, nausea, abdominal pain, chills, and fatigue. The acute reactions requiring intervention were managed by either temporarily interrupting or discontinuing infusion, and administering additional antihistamines, antipyretics, or corticosteroids.
To report SUSPECTED ADVERSE REACTIONS, contact BioMarin Pharmaceutical Inc. at 1-866-906-6100, or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Please see accompanying full Prescribing Information, including Boxed Warning.
VIMIZIM® (elosulfase alfa) is indicated for patients with mucopolysaccharidosis type IVA (MPS IVA; Morquio A syndrome).