





Of these, 36 received PALYNZIQ in Part 1:
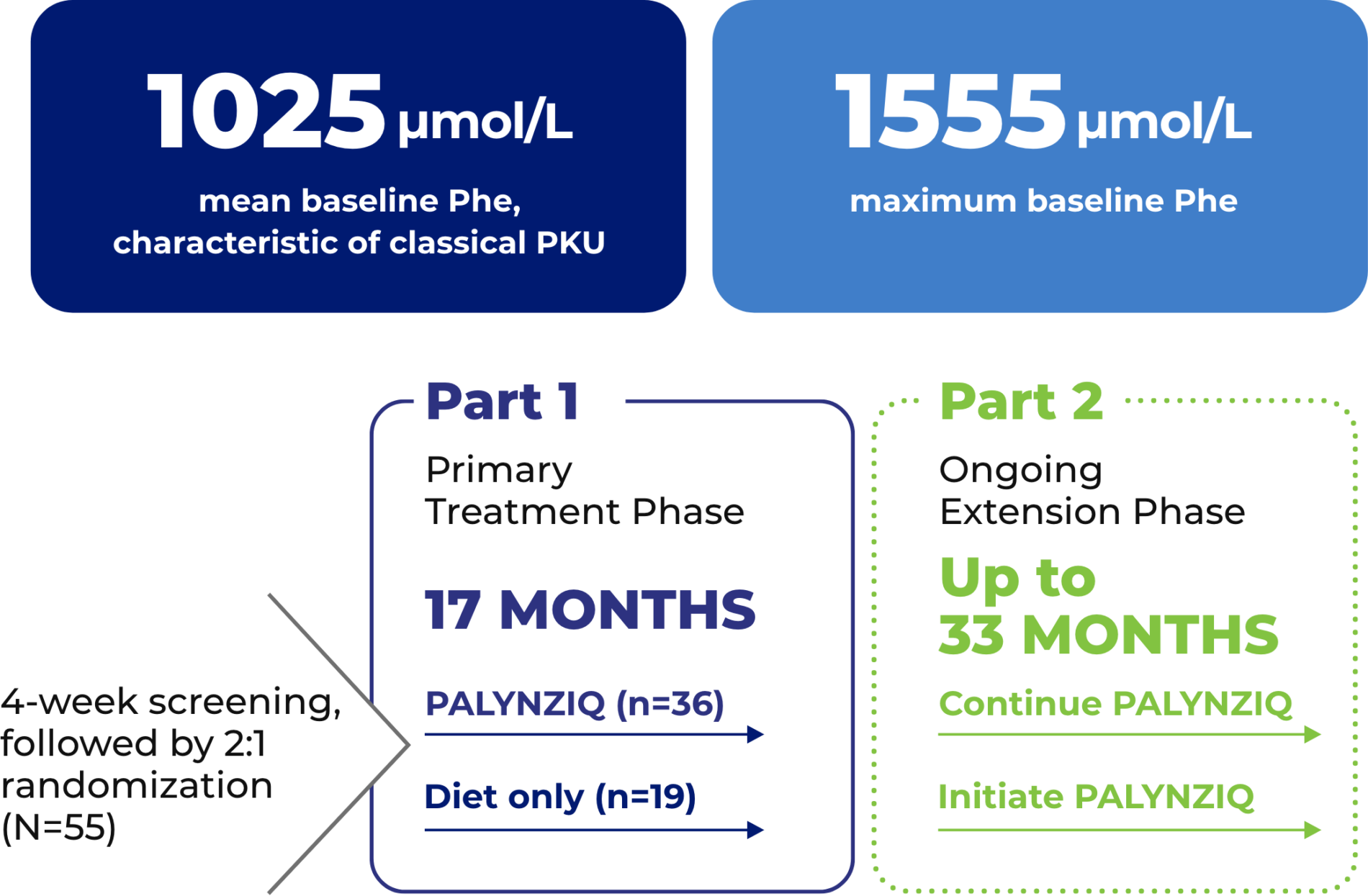

*The Phase 3 PRISM trial for PALYNZIQ enrolled 261 individuals regardless of baseline Phe or dietary Phe restriction; adherence to existing management did not exclude participation.1,2
†Patients aged 16-17 years were initially included in the trial prior to a protocol amendment.1,2
‡A Phe-restricted diet is defined as >75% of protein intake from medical food.1
§Patients were randomized 2:1 to either continue their randomized dosage of PALYNZIQ (20 mg or 40 mg once daily) or receive matching placebo.1,2
References: 1. PALYNZIQ package insert. Novato, CA: BioMarin Pharmaceutical Inc; February 2026. 2. Thomas J, Levy H, Amato S, et al, for the PRISM investigators. Pegvaliase for the treatment of phenylketonuria: results of a long-term phase 3 clinical trial program (PRISM). Mol Genet Metab. 2018;124(1):27-38. doi:10.1016/j.ymgme.2018.03.006 3. Data on file. BioMarin Pharmaceutical Inc. 4. Qi Y, Patel G, Henshaw J, et al. Pharmacokinetic, pharmacodynamic, and immunogenic rationale for optimal dosing of pegvaliase, a PEGylated bacterial enzyme, in adult patients with phenylketonuria. Clin Transl Sci. 2021;14(5):1894-1905. doi:10.1111/cts.13043 5. Harding CO, Longo N, Northrup H, et al. Pegvaliase for the treatment of phenylketonuria: final results of a long-term phase 3 clinical trial program. Mol Genet Metab Rep. 2024;39:101084. doi:10.1016/j.ymgmr.2024.101084 6. Sacharow S, Andrews A, Burrow TA, et al. Safety and efficacy of pegvaliase in adolescents with phenylketonuria: primary results from PEGASUS, a phase 3 open-label randomized controlled study. Presented at: International Congress of Inborn Errors of Metabolism (ICIEM) 2025; September 2-6, 2025; Kyoto, Japan.
BOXED WARNING: ANAPHYLAXIS
WARNINGS AND PRECAUTIONS
Anaphylaxis
Other Hypersensitivity Reactions
Injection Site Infections
Hypophenylalaninemia (HypoPhe)
Some patients have experienced HypoPhe; monitor blood Phe levels periodically during treatment. Frequent blood Phe monitoring is recommended in the pediatric population. For blood Phe concentrations below 30 micromol/L, the dosage of PALYNZIQ may be reduced and/or dietary protein and Phe intake may be modified to maintain blood Phe concentrations within a clinically acceptable range and above 30 micromol/L.
ADVERSE REACTIONS
The most common adverse reactions in clinical trials of primarily adult patients (at least 20% in either treatment phase) were injection site reactions, arthralgia, hypersensitivity reactions, headache, generalized skin reactions lasting at least 14 days, nausea, abdominal pain, vomiting, cough, oropharyngeal pain, pruritus, diarrhea, nasal congestion, fatigue, dizziness, and anxiety.
In the clinical trials, adverse reactions were associated with treatment discontinuation, dosage reduction and temporary drug interruption. In the 285 primarily adult patients exposed to PALYNZIQ in an induction/titration/maintenance regimen in clinical trials, 44 (15%) patients discontinued treatment due to adverse reactions.
Pediatric Patients: In a clinical study of 55 patients aged 12 to less than 18 years of age, the most common adverse reactions (at least 20% and greater than in control) were injection site reactions, arthralgia, headache, pyrexia, hypersensitivity reactions, dizziness, nausea, vomiting, fatigue, and pain in extremity. Two patients (5.6%) discontinued treatment due to adverse reactions.
Blood Phenylalanine Monitoring and Diet
DRUG INTERACTIONS
Effect of PALYNZIQ on Other PEGylated Products
USE IN SPECIFIC POPULATIONS
Pregnancy and Lactation
Available data do not establish an increased risk of adverse developmental outcomes to the fetus exposed to PALYNZIQ.
Pediatric & Geriatric Use: The safety and effectiveness of PALYNZIQ in pediatric patients from birth to less than 12 years have not been established. Clinical studies of PALYNZIQ did not include patients aged 65 years and older.
You are encouraged to report suspected adverse reactions to BioMarin at 1-866-906-6100, or to the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Please see full Prescribing Information, including BOXED WARNING, and the Medication Guide.
INDICATION
PALYNZIQ® (pegvaliase-pqpz) is a phenylalanine (Phe)-metabolizing enzyme indicated to reduce blood Phe concentrations in adult and pediatric patients 12 years of age and older with phenylketonuria (PKU) who have uncontrolled blood Phe concentrations greater than 600 micromol/L (10 mg/dL) on existing management.